|
Every iodine atom has 53 protons and the Z of iodine is 53.If an atom has one proton then the Z is 1 and the element is hydrogen.The Z is always determined by the number of protons. It does not matter how many neutrons and electrons are present in an atom. But, the valence electron determines the chemical bonding behaviour of an element. Helps in the determination of the properties of any element.The elements are arranged in order of increasing atomic numbers. Forms the basis of the arrangement of the elements in the periodic table.Helps in the identification of a particular element of an atom.You can download Atomic Number Cheat Sheet by clicking on the download button below However, with the addition of more number of protons and neutrons, the elements become prone to radioactive decay. Theoretically, atomic numbers can be increased if more elements are discovered. It starts with hydrogen and ends with the heaviest known element, Oganesson (Og). As a matter of fact, it ranges from 1 to 118. And protons are generally units of matter. Because it is the total number of protons. 
of Electrons”įurthermore, atomic numbers are whole numbers. This means that the number of electrons is equal to the number of protons present in an atom. We know that an atom consists of protons, neutrons and electrons. Each element is characterized by a unique atomic number. The total number of protons present in the nucleus of every atom of a chemical element represents the atomic number of that element. Towards Quantum Mechanical Model of Atom.Development Leading to Bohr’s Model of Atom.How are Electrons Distributed in Different Orbits (Shells)?.Browse more Topics under Structure Of Atom This number represents the number of protons in the nucleus of an atom. Moseley hypothesized the modern periodic table given by Mendeleev was on the basis of the number of protons in the nucleus of an atom.Īnd this hypothesis forms the basis of atomic number. Moseley in 1913 studied wavelengths of X-rays emitted by different chemical elements. It was not until Wilhelm Röntgen and his discovery of X-rays in 1895 that helped other scientists to further research on Mendeleev’s discovery. Some of the elements when arranged according to Mendeleev’s law were out of sequence. 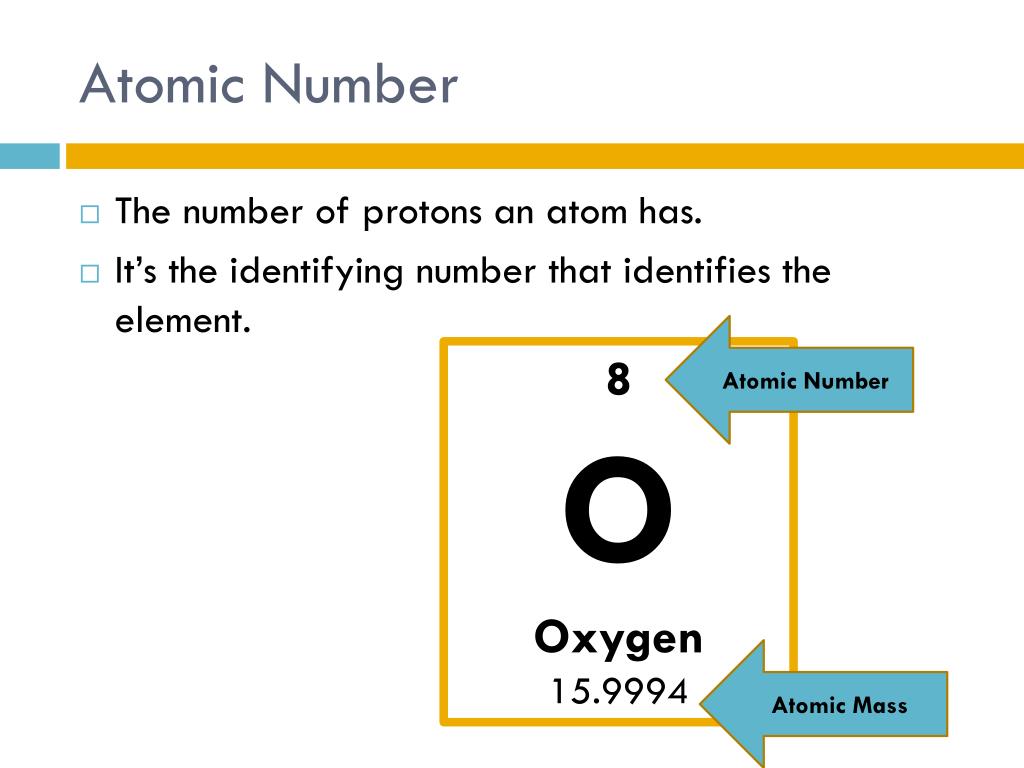
The periodic law soon became a major organizing concept of chemical sciences. In a time when Dmitri Mendeleev first discovered the periodic law. The history of periodic table dates back to the late 1860s. We all know that, in the periodic table, the elements are arranged in a very informative order.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
